The development of drugs that can be administered orally has always been a challenge in the pharmaceutical industry. Many proteins vital for treating various diseases have remained elusive to oral drug therapy, limiting patient convenience and accessibility. However, in a recent study published in Nature Chemical Biology, scientists from the laboratory of Professor Christian Heinis at EPFL have made a significant breakthrough in drug development. Their research has opened the door to a new class of orally available drugs, addressing a long-standing challenge in the field.
Overcoming the Limitations of Traditional Small Molecules
Traditional small molecules often struggle to bind to proteins with flat surfaces or require specificity for particular protein homologs. This limitation has hindered the development of drugs that can effectively target certain proteins. In particular, protein-protein interactions in cancers, which are crucial for tumor growth but cannot be inhibited, pose a major challenge for drug development. The study led by Professor Heinis and his team focused on addressing these limitations by exploring the use of cyclic peptides.
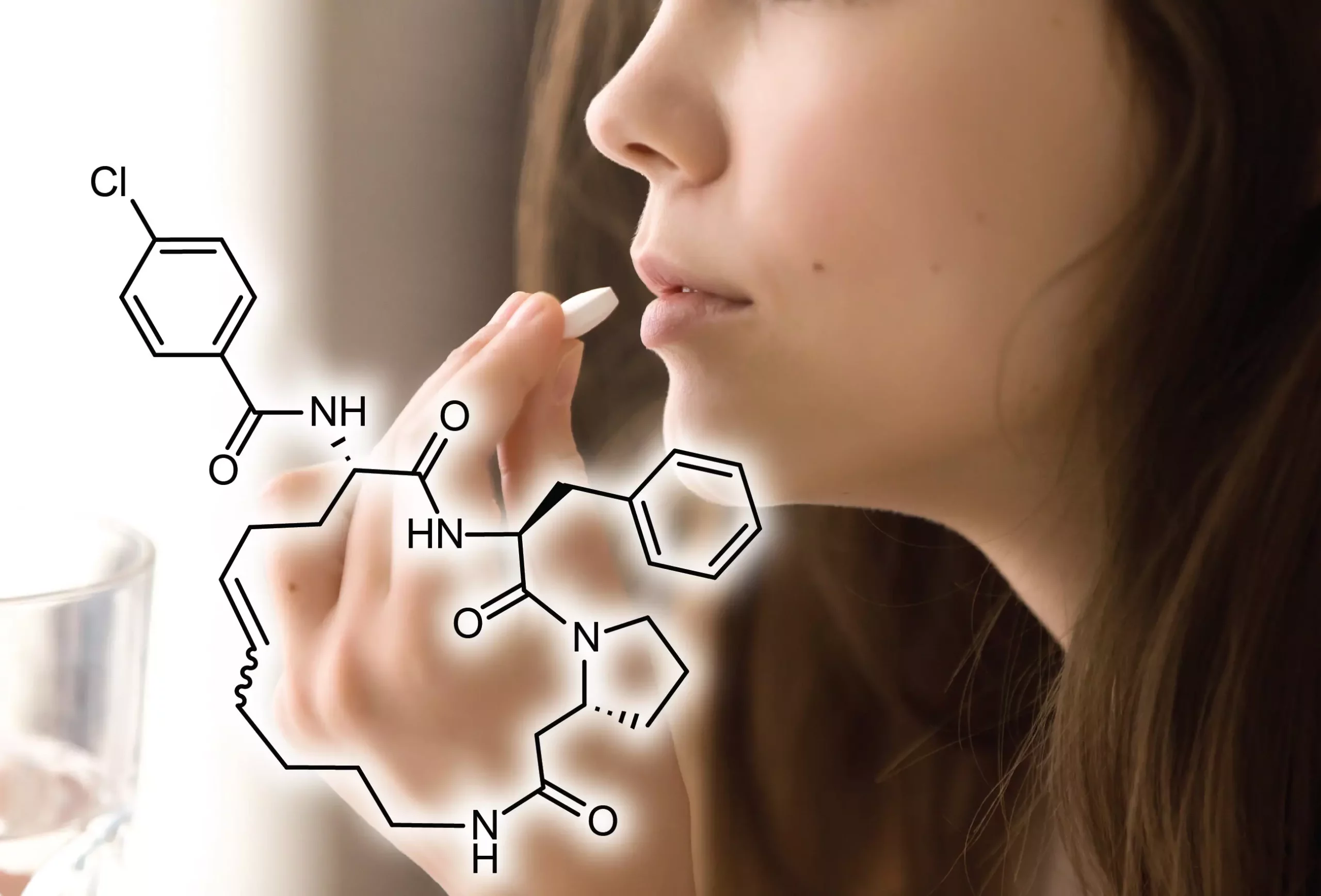
Cyclic peptides are versatile molecules known for their high affinity and specificity in binding challenging disease targets. These molecules have great potential for drug development, as they can bind to difficult targets that traditional methods have struggled to address. However, their application as oral drugs has been limited because they are rapidly digested or poorly absorbed by the gastrointestinal tract.
A Two-Step Combinatorial Synthesis Strategy
In order to develop orally available cyclic peptides, the scientists at EPFL developed a two-step combinatorial synthesis strategy. This novel approach allowed them to synthesize a vast library of cyclic peptides with enhanced metabolic stability when taken orally. The research team targeted the enzyme thrombin, which plays a critical role in blood coagulation and is key to preventing and treating thrombotic disorders such as strokes and heart attacks.
The first step of the process involved synthesizing linear peptides, which were then cyclized using bis-electrophilic linkers to form stable thioether bonds. In the second step, the cyclized peptides underwent acylation, resulting in the attachment of carboxylic acids and further diversifying their molecular structure. This method eliminated the need for intermediate purification steps and enabled high-throughput screening directly in the synthesis plates. Thousands of peptides were synthesized and screened to identify candidates with high affinity for thrombin.
The researchers successfully generated a comprehensive library of 8,448 cyclic peptides with an average molecular mass of about 650 Daltons (Da), slightly above the recommended maximum limit of 500 Da for orally-available small molecules. These cyclic peptides demonstrated a high affinity for thrombin. Additionally, when tested on rats, they showed oral bioavailability up to 18%, a substantial increase compared to the typical bioavailability of orally-administered cyclic peptides, which is below 2%.
By enabling the oral availability of cyclic peptides, the research team has opened up possibilities for treating a wide range of diseases that have previously been challenging to address with conventional oral drugs. This advancement is particularly significant for drugs in the biologics category, which includes peptides. The method’s versatility also allows for its adaptation to target various proteins, potentially leading to breakthroughs in areas where current medical needs are unmet.
In the next phase of the project, the researchers plan to target intracellular protein-protein interaction targets, which have been challenging to inhibit using classical small molecules. They are confident that the development of orally applicable cyclic peptides for these targets is within reach. The team also aims to automate further steps of the method, potentially enabling the synthesis and screening of libraries with more than one million molecules.
The research conducted by Professor Christian Heinis and his team at EPFL represents a significant advancement in drug development. The ability to create orally available cyclic peptides opens up new possibilities for the treatment of diseases that have been difficult to address with traditional oral drugs. This breakthrough has the potential to revolutionize the pharmaceutical industry and improve patient outcomes. With further research and development, orally available cyclic peptides may become a crucial tool in the fight against various diseases.

Leave a Reply