The University of Cincinnati’s team of engineers, led by Associate Professor Jingjie Wu, has made significant strides in the conversion of carbon dioxide into valuable products. This breakthrough not only addresses climate change but also offers a more efficient method for producing ethylene, a crucial component in the creation of various products, including plastics. With the ability to reduce carbon emissions and utilize renewable energy, this innovative approach holds great promise for a greener future.
Ethylene is widely regarded as one of the most essential chemicals globally due to its extensive usage across various industries. It serves as a key ingredient in the production of textiles, antifreeze, and vinyl. In 2022 alone, the chemical industry generated a staggering 225 million metric tons of ethylene. However, the conventional steam-cracking process used to produce ethylene contributes significantly to carbon dioxide emissions. By harnessing carbon dioxide as a feedstock instead of relying on fossil fuels, the team at the University of Cincinnati aims to revolutionize ethylene production, leading to a reduction in greenhouse gas emissions.
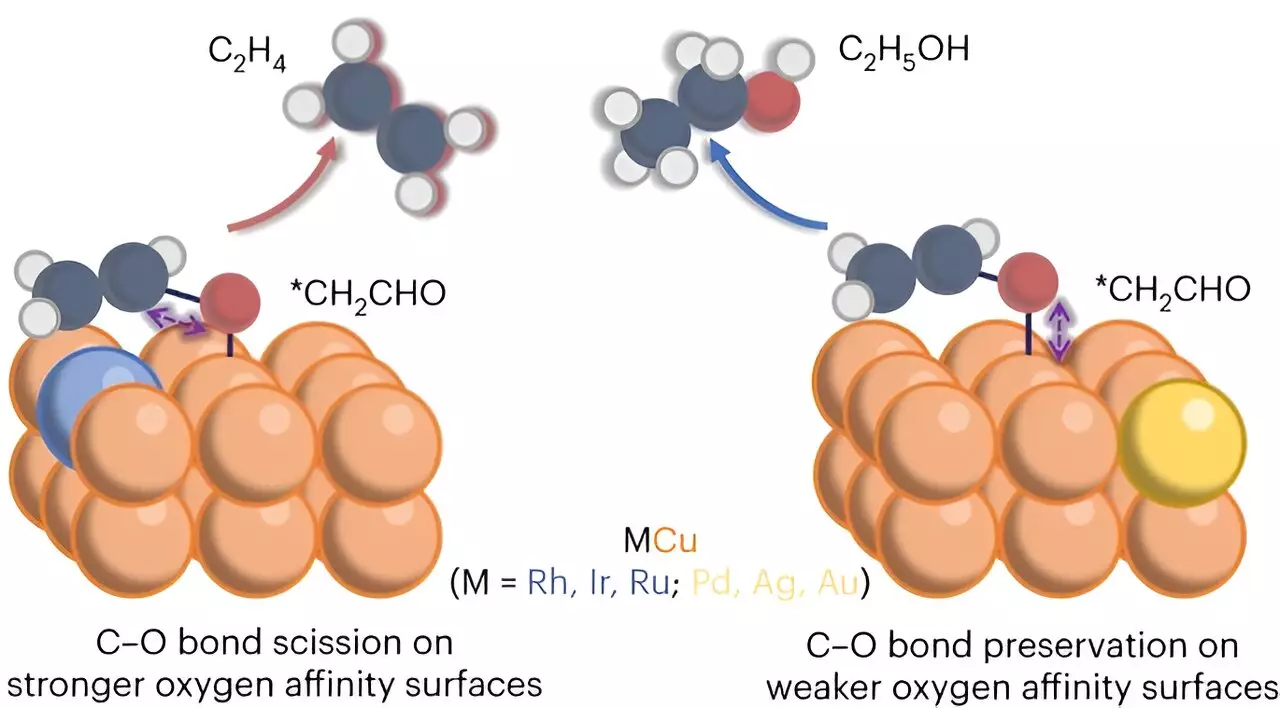
In their study published in Nature Chemical Engineering, Wu’s team, in collaboration with various institutions, including Rice University and Oak Ridge National Laboratory, focused on enhancing the electrochemical conversion of carbon dioxide into ethylene. Through the utilization of a modified copper catalyst, the researchers were able to enhance the production of ethylene. The team discovered critical insights into the divergence between ethylene and ethanol during the electrochemical CO2 reduction process, ultimately proposing a viable approach to directing selectivity toward ethylene. This breakthrough resulted in an impressive 50% increase in ethylene selectivity, marking a significant advancement in the field.
While the current process shows immense potential, there are challenges that must be overcome to transition it into commercial viability. As the conversion system operates, byproducts such as potassium hydroxide form on the copper catalyst, leading to reduced efficiency. To address this issue, the team acknowledges the need to enhance electrode stability and extend its operation from 1,000 to 100,000 hours. This refinement process is crucial for achieving the desired scalability and reliability necessary for widespread adoption.
Beyond the benefits specific to ethylene production, Wu emphasizes that these advancements in carbon dioxide conversion can significantly contribute to decarbonizing the chemical industry as a whole. By harnessing renewable electricity and sustainable feedstock, the industry can transition away from fossil fuels and embrace a greener and more energy-efficient future. Wu’s team believes that by integrating these new technologies, the chemical industry can make substantial progress toward sustainable practices, reducing its environmental impact and fostering a more sustainable and circular economy.
The breakthrough achieved by engineers at the University of Cincinnati paves the way for a more efficient and sustainable approach to carbon dioxide conversion. By focusing on the electrochemical conversion of carbon dioxide into ethylene, the team has demonstrated the potential to significantly reduce carbon emissions and reliance on fossil fuels. With ongoing efforts to enhance the process’s commercial viability, this research holds immense promise for transforming the chemical industry, making it greener and more energy-efficient. Through innovative solutions like these, we move closer to a sustainable future where environmental preservation and technological advancements go hand in hand.

Leave a Reply