The search for effective treatments to block the growth of cancer cells is an ongoing challenge in the field of oncology. Tumor cells rely on thousands of proteins to function, but only a few of these proteins can be targeted by drugs safely and effectively. However, a recent study conducted by a collaborative team at Scripps Research and the Broad Institute of Harvard and MIT has introduced a novel method to identify new drug targets that have the potential to impact multiple types of cancer. This groundbreaking research, published in Nature Chemical Biology, utilized a precise gene editing technique to alter over 13,000 possible drug targets and determine which alterations affected cell growth. By integrating this information with chemical proteomic data, the team identified hundreds of potential drug targets, many of which have not been explored in previous studies.
The approach used in this study combines two innovative methods to create a comprehensive strategy for identifying new cancer treatments. Benjamin Cravatt, Ph.D., the Norton B. Gilula Chair of Chemical Biology and chemistry professor at Scripps Research, explains that this method provides valuable preclinical information about the specific protein sites that are most likely to impact cancer cell growth. This integrated approach has the potential to revolutionize the way drug targets are identified and facilitate the development of more effective cancer treatments.
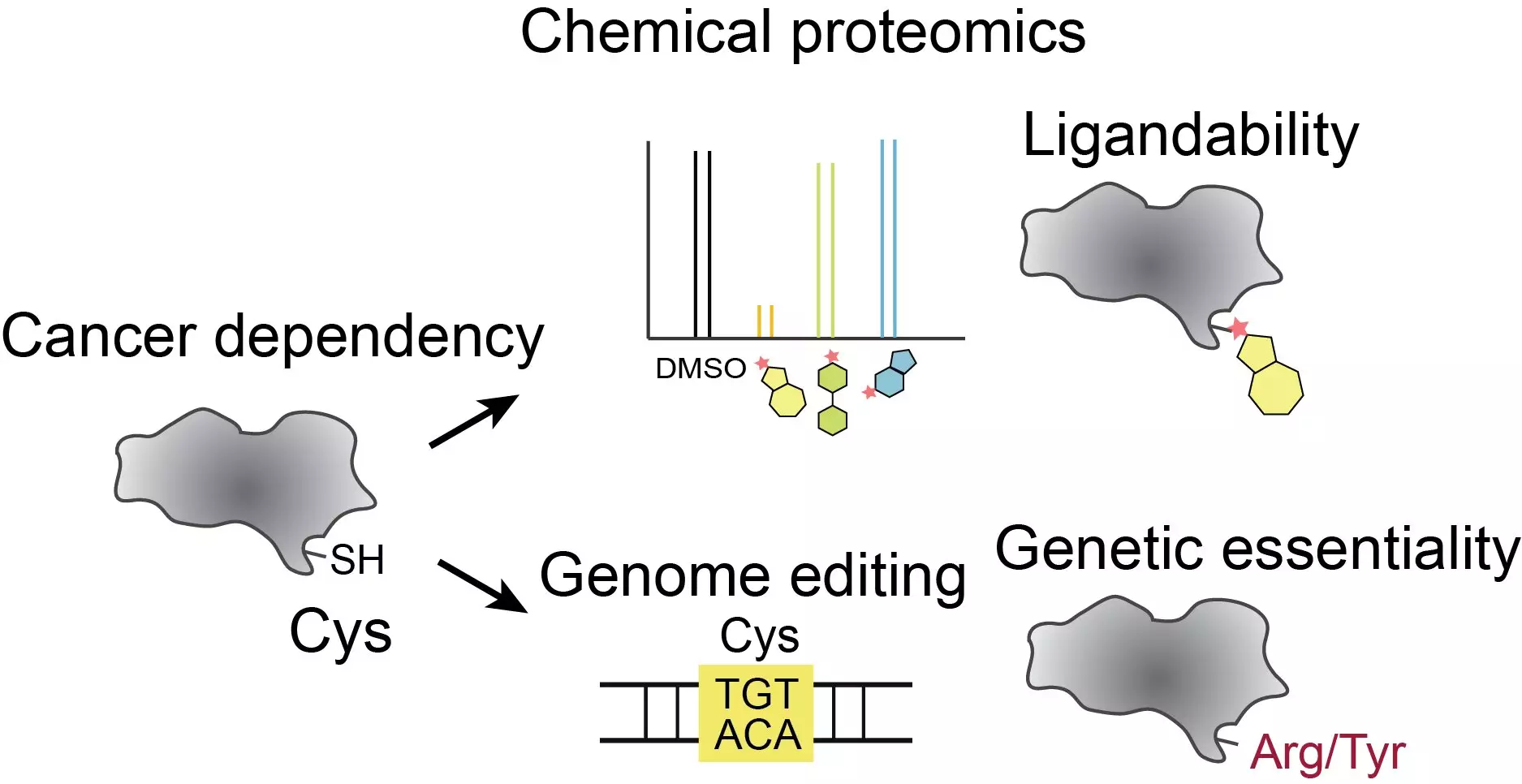
One of the major challenges in cancer research is identifying which proteins should be targeted with drugs. A particular focus has been on cysteines, one of the twenty amino acids that make up human proteins. Cysteines have unique reactive chemistry, making them ideal drug targets. However, with hundreds of thousands of cysteines scattered among human proteins, narrowing down the specific ones that play a crucial role in cancer cell growth is extremely difficult. Even among the few thousand proteins that have been identified as critical to cancer cell growth, there are still over 13,000 cysteines. The researchers needed a method to pinpoint the cysteines that have the greatest functional impact on cancer-relevant proteins.
To address this challenge, the researchers combined precise genome engineering techniques with cutting-edge chemical proteomic tools. They utilized base editing, a technique developed by the lab of David Liu at the Broad Institute, to create targeted amino acid changes in cancer cells. By introducing various cysteine-targeted mutations, they aimed to understand which cysteines were most relevant to cancer cells. The team edited 13,800 spots on more than 1,750 genes previously linked to cancer cell survival, specifically targeting cysteines on the corresponding proteins. They then assessed how well the cancer cells with these mutations grew. Furthermore, they integrated their findings with new data on the “druggability” of these cysteines.
The study revealed that approximately 160 of the druggable cysteines, when edited, had a significant impact on cancer cell growth, indicating that drugs binding to these cysteines could potentially be used to treat cancer. One of the most notable findings was the change made to the cancer-dependency protein TOE1. While TOE1 is known to play a role in modifying the ends of the cell’s RNA molecules, it had not been previously studied as a target for cancer treatment. However, the researchers demonstrated that small molecules could be used to target TOE1 effectively. This finding opens up new possibilities for developing drugs that can block the normal activity of TOE1 and potentially inhibit cancer cell growth.
Although further research is needed to determine if drugs targeting TOE1 could be effective in human patients, the initial results of this study are highly promising. The method of editing cysteines proved to be successful in predicting which cysteines could serve as dru

Leave a Reply